What Is The Electromagnetic Spectrum?
The whole range of electromagnetic waves, from visible light all the way up to gamma rays, is referred to collectively as the em spectrum. Understanding this region of the globe is crucial, and this topic is an essential component of the scientific discipline. The following paragraphs will explain how many of the most important characteristics of this spectrum may be used.
The following categories make up the electromagnetic spectrum:
- Radio waves: Radio waves have the longest wavelengths of all the electromagnetic waves, yet their frequencies and energy are the lowest. Both communication and navigation rely on them, as seen by the prevalence of radio and television broadcasts, as well as the Global Positioning System (GPS).
- Microwaves: Microwaves are distinguished from radio waves by their shorter wavelengths, higher frequencies, and greater energy. They play a role in communication, such as in cellular phones and the transmission of satellite signals, as well as in the preparation of food in microwave ovens.
- Infrared radiation: Compared to microwaves, infrared radiation has shorter wavelengths as well as greater frequencies and energy. It is detectable as heat and is given out by things that are already heated. Thermal imaging cameras and systems for remote control both make use of infrared radiation as an image source.
- Visible light: Light that can be seen by the naked eye is referred to as visible light. Visible light refers to the fraction of the electromagnetic spectrum that can be seen by humans. The wavelengths of this kind of radiation are shorter than those of infrared radiation but longer than those of ultraviolet light. That region of the spectrum that contains the colors of the rainbow is referred to as "that portion."
- Ultraviolet radiation: Ultraviolet radiation is distinguished from visible light by its shorter wavelengths, higher frequencies, and more intense energies. It may cause sunburn and is damaging to the living tissues it comes into contact with. Tannin' beds and lights designed to kill germs both make use of it.
- X-rays: In comparison to ultraviolet light, X-rays have shorter wavelengths while also having greater frequencies and energy. They are used in imaging devices, such as X-ray machines, which are used to see the inside of the body.
- Gamma rays: Gamma rays are distinguished from other forms of electromagnetic radiation by possessing very short wavelengths in addition to extremely high frequencies and energy. Radioactive materials give out these particles, which have the potential to cause damage to living tissue. In addition to being used to sterilize medical equipment, they are employed in the treatment of cancer.
Infrared In The electromagnetic Radiation Spectrum
The portion of the electromagnetic spectrum of radiation that extends beyond the end of the visible light spectrum that is designated as infrared is known as infrared. The infrared band is used for the purpose of determining the thermal characteristics of various things. In addition to that, it is used in night vision apparatus.
Radiation that is considered to be infrared has a frequency that may vary anywhere from approximately 300 gigahertz to 400 terahertz and a wavelength that can be anywhere from about one millimeter to one micrometer. The heat that we experience is a result of a certain form of electromagnetic radiation. Radiation in the infrared spectrum may be put to work for a number of different tasks, including sensing, heating, and cooking. Buildings and outdoor areas may both benefit from the warmth that infrared heaters provide. Temperature readings may be taken using infrared cameras, and they can provide clear images in the dark.
Wavelengths of infrared radiation are far more compact compared to those of microwave or radio radiation. They have a unit of measurement known as microns. Despite their shorter length, these wavelengths have the potential to cause damage to the human body.

Near infrared and far infrared are the two primary subcategories that make up the infrared spectrum. The near infrared is the range of wavelengths that include the frequencies that are the shortest. The length of these wavelengths falls anywhere between one and five microns. Additionally, there are bands that are intermediate and lengthy in the infrared spectrum. Each is distinguished by the particular wavelengths that it has.
The most well-known use of infrared technology is in night vision goggles worn by military personnel. When worn at night, these goggles convert infrared light into wavelengths that are visible to the human eye. Infrared light, on the other hand, is used in both wireless and wired forms of communication.
Comprehending The Electromagnetic Spectrum
A range of adverse health consequences, including hyperthermia, may be brought on by prolonged exposure to infrared light. The rate of damage is proportional to the strength of the exposure as well as the length of time it is received. One of the symptoms is a rapid and irreversible rise in the temperature of the skin, which, if left untreated, may cause irreparable damage.
Because infrared light may enter the human eye, the cornea is particularly vulnerable to damage caused by heat. Cataracts may result from this condition if left untreated.
The temperature of objects, like as stars and planets, may also be determined with the use of infrared technology. In addition to that, it may be used to identify molecular clouds. Other uses include those in the medical field, in telecommunications, and in communications using fiber optics.
The International Commission for the Protection against Ionizing Radiation (ICNIRP) has published recommendations for the limits of exposure to incoherent visible and infrared radiation.
Light that may be seen on the electro magnetic spectrum
The electromagnetic specturm includes the range of wavelengths that may be seen as visible light. The sun is the primary source of light that we have. The moon and the stars are two more sources of light that are visible to the human eye. It is essential to be aware that our eyes are unable to detect infrared and ultraviolet wavelengths of light. Nevertheless, we are able to distinguish between the red and blue light. The culmination of these color contributions is what we refer to as white light.
The kind of electromagnetic radiation that humans are able to see is known as visible light. Its frequency may be anywhere from approximately 400 terahertz to about 750 terahertz, and its wavelength can be anywhere from about 400 nanometers to about 750 nanometers. There are many applications for visible light, including illumination, communication, and sensing, and these are only a few of them. LEDs, which stand for light-emitting diodes, are used for the purpose of illumination, whilst fiber-optic cables are utilized for the purpose of communication. Visible light may be detected by both cameras and the human eye.
The idea that light is made up of a collection of rays is connected to this idea, as well. A prism is used in order to accomplish the task of separating these photons into their respective spectral hues. A device known as a prism is one that may deflect a ray of light. This is the same technology that is used for the production of cellular telephones.
The electromagnetic spectrum of colors that light may appear as is one of the most fascinating elements of light. Colored light comes in a variety of colors, the most common of which are red, blue, and green. Other colors include yellow, orange, and violet in addition to the more common red, blue, and green. It's fascinating to think that a single prism could split all of these colors into their own hues, creating a rainbow. A fascinating experiment in physics may be carried out by dividing light into its many spectral bands using a prism.
The ems spectrum also consists of many other, less well-known components, such as infrared and radio waves, for example. Some of these have been put to use in radio, television, and mobile phone transmissions. However, the best method to make advantage of them is to develop the appropriate sort of filter for the situation. We can lessen the toll that these components have on our bodies if we choose this course of action. In a similar vein, we are able to construct a virtual setting in which it is risk-free for us to investigate these components, and we can do so even without using our actual eyes.
Even though the smallest and longest wavelengths of visible light may be the most noticeable, the shortwave infrared (SWIR) and microwave frequencies are the waves that have the highest energy efficiency and have the most visually pleasant appearance.
UV Emr Spectrum
Radiation known as ultraviolet (UV) is a portion of the spectrum of electromagnetic energy. It is adaptable, and may be used in a variety of contexts. But it also has the potential to be dangerous. Radiation with UVB and UVC wavelengths is harmful to human eyes and may also lead to diseases of the skin.
Radiation in the ultraviolet spectrum has a frequency that may vary from approximately 750 terahertz to 30 picohertz and a wavelength that can range from roughly 10 nanometers to 400 nanometers. Sunburns are brought on by a specific kind of electromagnetic radiation called ultraviolet radiation. Ultraviolet light has a wide range of applications, some of which include sensing, sterilizing, and tanning. UV lamps are used for the purpose of surface sterilization and tanning beds, whereas UV sensors are utilized for the purpose of monitoring UV radiation.
This kind of energy may be taken up by molecules, which can then kickstart various chemical processes. The molecule that absorbs light may then either fluoresce or emit visible light.
There are three primary subgroups that make up the UV spectrum; these are the extreme, the close, and the distant regions. Arc lamps, lasers, and light-emitting diodes are three examples of common types of UV sources.
UV photons have a higher energy level than X-rays despite having shorter wavelengths. This is because UV rays have shorter wavelengths. In certain cases, this may be helpful in severing chemical connections. The term "nonionizing radiation" may also be used to refer to these waves.

In biochemistry, one of the most common methods for determining how well a material absorbs radiation is to utilize the UV spectrum. There are many different kinds of compounds, each with its own unique pattern of substantial light absorption bands in the UV.
Spectroscopy often makes use of a light that is stable against UV radiation. Argon and deuterium arc lamps are popular sources.
Certain types of insects have a remarkable capacity for detecting UVB radiation. Birds are equipped with a fourth color receptor, which allows them to distinguish UV light. In addition to that, they may utilize UV to locate their prey.
The sun is the source of ultraviolet light, which is a kind of electromagnetic radiation and belongs to the spectrum. Its frequency range is from 800 terahertz to 30 picohertz, and its range of measurement is between ten and four hundred nanometers. However, the majority of individuals are unable to see it.
One explanation for this is because the human eye does not include any color receptors. However, the UV range is essential for the survival of many creatures. For instance, the human body is unable to create natural vitamin D without UVB's assistance. It is used by other animals as a means of locating their prey.
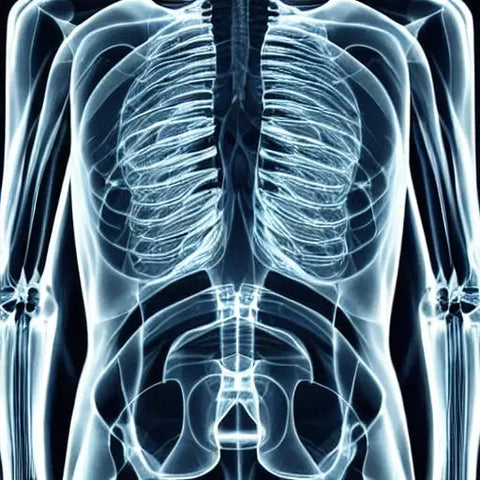
X-rays
High-energy electromagnetic radiation is what we refer to as x-rays. In contrast to gamma rays and ultraviolet light, X-rays have shorter wavelengths than visible light and are thus able to pass through materials that are quite thin. They have a range of uses in medicine, such as imaging bones and tissues, among other things. There is more than one kind of X-ray.
X-rays have a frequency that can vary anywhere from 30 pHz to 30 eHz and have a wavelength that can range anywhere from 0.01 nanometers to 10 nanometers. They belong to the category of electromagnetic radiation and have the ability to pass through solid materials like bones. Imaging patients with X-rays and inspecting items for flaws in their metal components are just two of the many medical and industrial applications for this technology.
Low-energy photons interacting with a magnetic field may result in the production of nonthermal X-rays. Thermal X-rays are created when a hot gas is used. In addition to their usage in medical imaging, these devices also find use in industrial radiography, scanners used in airport security, and quality control for a variety of materials.
The collision of an incoming electron with an atom results in the emission of hard X-rays. Because of this, there will be an empty spot in the atom's electron shell. There is a chance that a second electron will occupy the vacuum. An other possibility is that the approaching electron will dislodge an atom. When anything like this takes place, some of the energy contained inside the photon is passed on to the electron that is scattering it.
Less than 10 nanometers is the minimum wavelength for soft X-rays. These are used in the field of X-ray microscopy, where they are utilized to collect pictures of the interiors of various tiny structures, including biological cells.
One of the four possible nuclear processes may result in the emission of hard X-rays. Ionization, Compton scattering, and photoabsorption are the three primary varieties of interactions. At higher energy, the Compton scattering process is the dominant one. At lower X-ray energy, the most prevalent kind of interaction is called photoabsorption.
It is important to note that an X-ray is not the same thing as the X-band, which is a low-energy portion of the electromagnetic spectrum. Despite the fact that the two bands overlap one another by a few hundred nanometers, they do not share the same properties.
Since X-rays are capable of penetrating objects, they may be used in a wide number of contexts. During the security screening procedure, for instance, X-rays are used to check for fractures in the luggage. In addition to that, patients undergoing radiation for cancer are given these. In addition, X-rays may be utilized to analyze the structural components of various materials, such as cement.
Gamma rays
Electromagnetic radiation may come in a variety of forms, one of which is the gamma ray. In point of fact, gamma rays are comprised of all photons with extraordinarily high energies. These photons are created as a byproduct of nuclear disintegration as well as other high-energy physics investigations. Within the electromagnetic spectrum, these photons have the highest kinetic energy.
The wavelength of gamma rays spans from around 0.001 nanometers to 0.01 nanometers, and their frequency ranges from approximately 30 EHz to 30 PHz. They belong to the category of electromagnetic radiation and have a very high energy level, making them potentially harmful to all forms of life. Gamma rays have several applications, including diagnostic imaging and therapy in the medical field. They are also used in a number of manufacturing processes.
Gamma rays are able to go quite far through materials due to the high amount of energy that they possess. Gamma rays have the potential to pass through many inches of lead without being stopped.
Radioactive elements' nuclei are the source of gamma rays with the highest energy levels. Gamma rays, on the other hand, are created when subatomic particles interact with one another.
Nuclei may be affected by the actions of particles such as electrons, muons, and pions. They are able to investigate the distribution of charge inside the nucleus. There is also the possibility of a pion engaging in a process of double charge exchange with the nucleus.
Gamma rays are produced as a byproduct of a number of high-energy physics experiments. For instance, a stream of relativistic particles that has been focussed by the magnetic field of a hypernova may be detected 10 billion light years away from the hypernova.

After going through the process of radioactive decay, the nucleus of certain radionuclides will give out gamma rays. Atomic transitions, annihilation, and interactions between subatomic particles are some examples of other sources of gamma rays.
The vast majority of gamma rays in astronomy may be traced back to other types of processes. The most powerful types of electromagnetic radiation are gamma rays, which may be produced by supernovae as well as nuclear fallout. As a result, they are an excellent resource for discovering new parts of the cosmos.
It's possible that exposure to certain gamma rays might harm the body's cells. Gamma rays, on the other hand, have a lower rate of ionization compared to beta and alpha rays, which means they have a lower risk of causing cancer. Gamma rays, despite this, have the potential to alter the structure of DNA and to cause burns. Ionization may be caused in the body by even the most minute amounts of gamma ray exposure.
Microwaves
Microwaves are a kind of electromagnetic radiation that have a frequency that may vary anywhere from 300 megahertz to 300 gigahertz and have a wavelength that can be anywhere from one millimeter to one meter. They provide a number of functions, including those related to communication, navigation, and heating, among others. Cell phone networks, Wi-Fi networks, and satellite communications all make use of microwave technology. They also play a role in radar and the process of food preparation in ovens.
Radio Waves/Signals
The kind of electromagnetic radiation with the longest wavelength and the lowest frequency is called radio waves. They have a frequency that may be anywhere from three kilohertz to three hundred gigahertz, and their wavelength can be anywhere from one millimeter to one hundred kilometers. Radio waves have a wide range of applications, some of which include communication, navigation, and radar systems. They are necessary for the transmission of signals for cellular phones, Wi-Fi networks, radio and television transmissions. They are also used for navigation, such as in GPS systems, and for radar, which is employed in weather forecasting and aircraft navigation. Both of these applications need accurate positioning and orientation.




















































Leave a comment